
Imaging Facility
Bringing together the microscopes of The ACRF Telomere Analysis Centre (ATAC) and other microscope and image analysis technologies.
Advanced Microscopy (AMC) aims to enable world-leading research by providing comprehensive support for light microscopy techniques. The centre supports experimentation from conceptual planning to data analysis and publication through training, guidance, and instrument access.
Eleven light and laser-based microscopes are available to support techniques including fluorescence microscopy, live-cell imaging, confocal microscopy, super-resolution microscopy, lightsheet imaging, and stereomicroscopy. Seven workstations are available for image analysis and data visualisation.
Explore More
Techniques
Advanced Microscopy Centre Techniques
Fluorescence Microscopy
Fluorescence microscopy is a workhorse technique for localizing cellular components. Our Zeiss AxioImager Microscopes give researchers advanced widefield fluorescence capabilities including Z-stacks, tiling, and ApoTome imaging. Our Zeiss AxioImager microscopes with MetaSystems Metafer software enable researchers to automate imaging and analysis of metaphase chromosomes and interphase nuclei.

Live Cell Imaging
Viewing cells in their living state provides a wealth of information not available from fixed specimens. Live cell imaging involves the collection of images at successive time intervals under conditions that support cell culture. Four of our microscopes support live cell imaging: a Zeiss Cell Observer for transmitted light and widefield fluorescence imaging, a Zeiss Cell Observer Spinning Disc confocal and Zeiss LSM 880 Airyscan for confocal and super-resolution imaging and a Leica Stellaris 8 FaLCon STED for confocal, Lifetime and super resolution imaging of live cells.
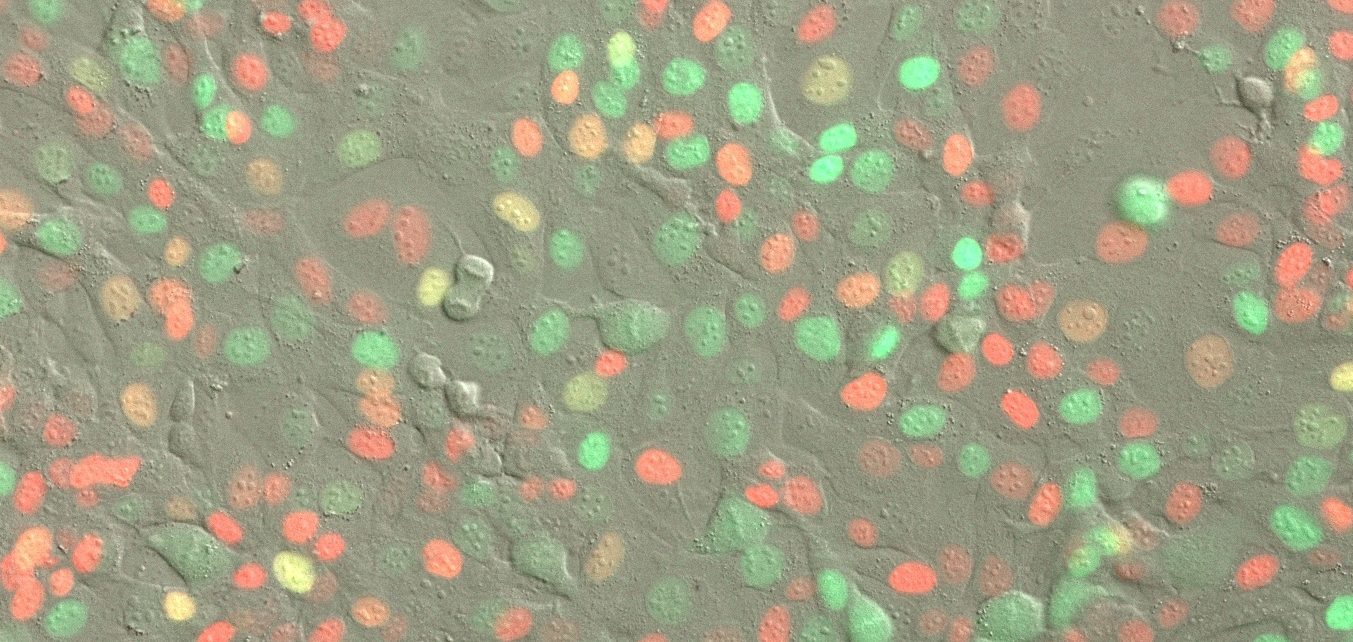
Confocal Microscopy
Confocal microscopy enables researchers to visualize cellular structures in three dimensions. Our laser-scanning confocal microscope, Zeiss LSM 880 Airyscan, has seven laser lines, selectable emission range and spectral unmixing capabilities. The Zeiss Cell Observer SD microscope supports multichannel confocal imaging of live cells using spinning disk technology. Our Leica Stellaris 8 FaLCon STED has a supercontinuum white light laser providing pulsed, tuneable excitation energy from 440-790nm and 5 hybrid (3x HyD S, 1x HyD X, 1x Hyd R) detectors spread across 400-800nm.

Super-Resolution Microscopy
Visualising separate objects smaller than the theoretical limit of optical resolution is called super-resolution.
With the Zeiss LSM 880 Airyscan, standard fluorescent samples and live cells can be imaged with as low as 120 nm resolution.With the Leica Stellaris 8 FaLCon STED using a 775nm depletion laser and lifetime-gating (Tau-STED) fluorescent samples can be imaged with as low as 30nm (x&y) by 100nm (z) resolution.

Lightsheet Imaging and Stereomicroscopy
Lightsheet imaging is an optical microscopy technique in which a section of a sample is illuminated with a thin sheet of light, resulting in robust, fast and gentle three-dimensional imaging of large specimens. When combined with a tissue clearing sample preparation technique, large tissue samples or whole-mount embryos up to 5 mm thick can be imaged using the Zeiss Lightsheet Z.1/7 microscope. Optically sectioning fluorescent samples using Lightsheet microscopy can replace traditional serial sectioning in a fraction of the time while retaining the overall three-dimensional structure of the sample. Using a more traditional microscopy technique, the Zeiss SteREO Lumar.V12 Fluorescence Stereomicroscope enables imaging large samples such as embryos, zebrafish and whole tissues.

Instruments
Advanced Microscopy Centre Instruments

Zeiss Lightsheet Z.1/7 Microscope
The Zeiss Lightsheet Z.1/7 is a fluorescent microscope designed to image larger specimens ranging in size from 0.5 mm to 5 mm. Specimens are able to be rotated 360 degrees, allowing the entire specimen to be imaged from different views before being digitally reconstructed. We have 5x and 20 objectives and specialized sample chambers are available for live samples or samples that have been cleared using certain tissue clearing techniques, including SCALE. The Lightsheet Z.1/7 is accompanied by a specialised image analysis workstation.

Zeiss SteREO Lumar.V12 Fluorescence Stereomicroscope
The Zeiss SteREO Lumar.V12 stereomicroscope is capable of high-resolution three-dimensional imaging over a large field of view. This makes it the ideal system for imaging larger samples such as embryos, zebrafish and whole tissues. It has brightfield reflected and transmitted light capabilities, as well as a range of filters (DAPI, GFP, YFP, RFP) allowing fluorescence imaging. The Zeiss SteREO Lumar.V12 stereomicroscope is a fully motorised with a magnification range from 9.6x to 120x and is capable of multichannel acquisition, Z-stacks, time lapse and tiling experiments.
Zeiss LSM 880 Airyscan Laser Scanning Confocal Microscope
Laser scanning confocal microscopy is an essential method in cell biology research for its ability to optically section and reconstruct cellular samples in three dimensions. The Zeiss LSM 880 is equipped with the Airyscan detector. This microscope allows visualization of intracellular structures in higher resolution, up to the super-resolution level.

Zeiss Cell Observer SD Spinning Disk Confocal Microscope
The Zeiss Cell Observer SD microscope uses Yokagawa spinning disc technology to generate 3D confocal images extremely quickly.

Zeiss Cell Observer Widefield Live Cell Microscope
Visualizing living cells offers a dimension to experiments that fixed (preserved) specimens can’t provide. Live cells can be imaged over the course of multiple hours or days. Cells can be imaged unlabelled or tagged with fluorescent proteins.

Zeiss Axio Imager Z2 with ApoTome.2 Widefield Upright Microscopes
Our two Axio Imager upright fluorescence microscopes are the ‘workhorses’ of the facility, allowing researchers to capture images of a wide range of fluorescently-labelled cell samples. The ApoTome.2 instrument allows the reconstruction of images in three dimensions with enhanced resolution and contrast.
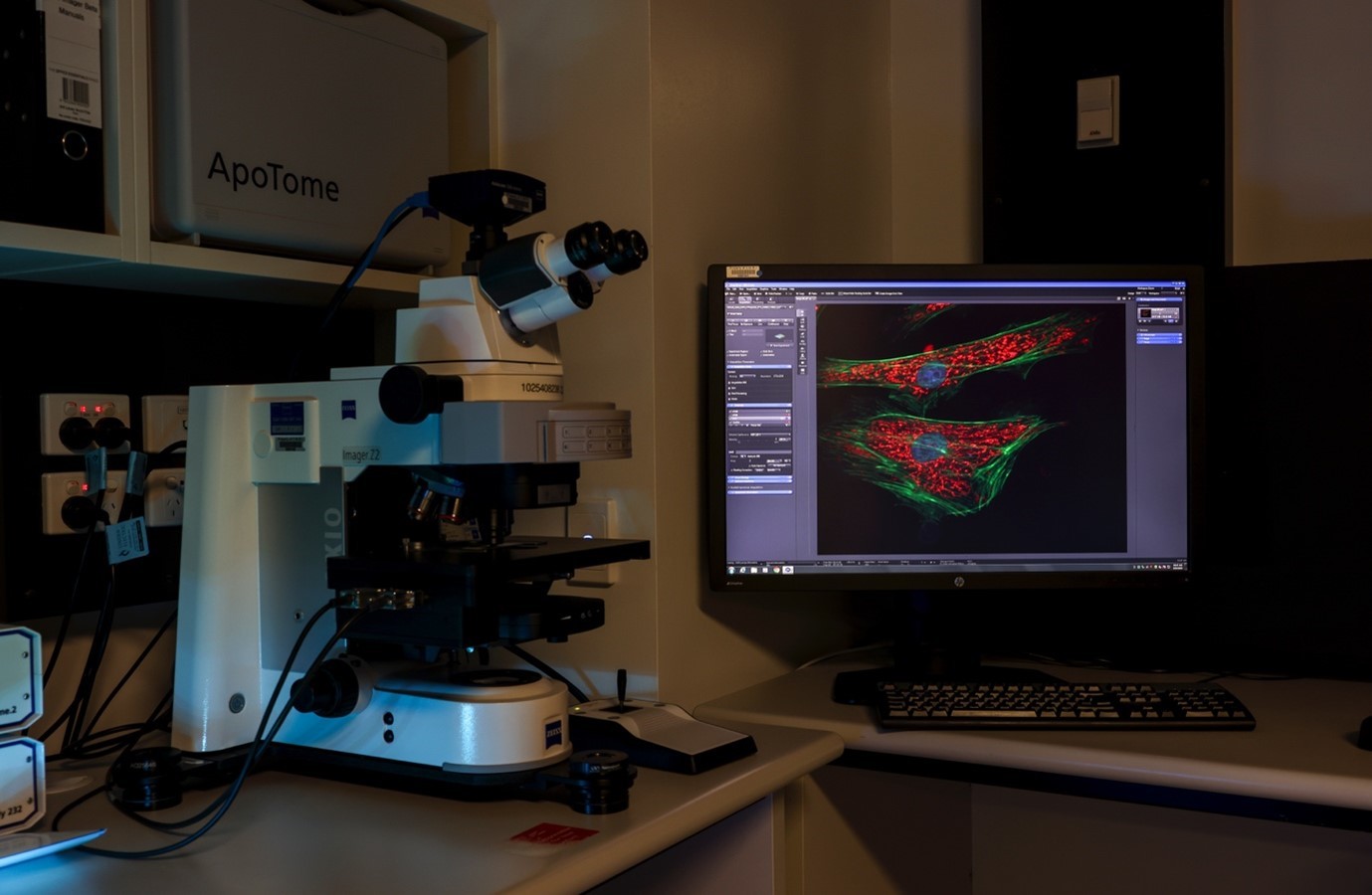
MetaSystems Metafer Chromosomal Analysis Microscopes
CMRI researchers pioneered the use of automated metaphase chromosome imaging in telomere research using the Metafer automated fluorescence microscopy system. Our two Metafer systems accelerate research by automating fluorescence microscopy of cancer and normal cells, enabling imaging in a few hours cell that would otherwise have taken weeks to complete.

Leica Stellaris 8 FaLCon STED
Our Leica Stellaris 8 FaLCon STED is a fully integrated system enabling confocal, fluorescent lifetime and super-resolution imaging in fixed or live samples.
It has a 405nm and supercontinuum white light laser 440-790nm and 5 hybrid (3x HyD S, 1x HyD X, 1x Hyd R) detectors spread across 400-800nm.
Fluorescent lifetime imaging by FaLCon, Fast Lifetime Contrast is a fully integrated form of time correlated single photon counting using the WLL and HyD detectors.
Super Resolution Imaging is possible in fixed and live samples using Tau-STED, lifetime-gated stimulated emission depletion microscopy using a 775nm depletion laser. The spatial resolution of Tau-STED is about <30 nm for fixed specimen and <70 nm for live specimens compared to 200 nm in a regular confocal microscopy.

The ACRF Telomere Analysis Centre
The Australian Cancer Research Foundation Telomere Analysis Centre (ATAC) was opened in 2015 as Australia’s first centre dedicated to supporting research into the cellular functions of telomeres.
Telomeres, nucleoprotein structures that protect chromosome ends, have emerging roles in cancer risk and oncogenesis, and the telomere maintenance mechanisms that are activated in almost all cancers are prime targets for the development of new cancer diagnostics and therapeutics.
The primary scientific focus of ATAC is centred on the study of telomeres and their roles in cell proliferation, cancer and ageing, with the secondary focus to support a broad range of medical and biological research projects. ATAC was developed by Kids Cancer Alliance telomere research leaders from Children's Medical Research Institute, Sydney Children's Hospital Network, and Children's Cancer Institute. The centre was established with the support of the Australian Cancer Research Foundation (ACRF) and the Ian Potter Foundation. ACRF granted $2 million to Children’s Medical Research Institute (CMRI) to fund equipment for the centre, which is located in a purpose-built facility within CMRI's building expansion at Westmead.
Read the ATAC 2020 Annual Report: Download PDF
Supported by:

Also supported by:















